Abstract
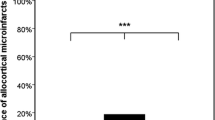
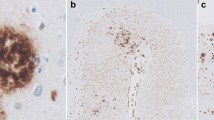
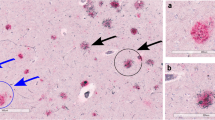
Recent studies reported both positive [Thal et al. (2003) J Neuropathol Exp Neurol 62:1287–1301] and negative [Tian et al. (2003) Neurosci Lett 352:137–140] correlations between cerebral amyloid angiopathy (CAA) and Alzheimer’s disease (AD) pathology. We have recently shown high correlations between neuritic AD pathology and amyloid β peptide (Aβ) deposits in the capillary/pericapillary compartment (CapCAA) with only low correlations to general CAA (non-capillary). We have now studied the relationship between CapCAA and AD pathology with respect to the distribution of Aβ40 and 42 in the frontal cortex of 100 human postmortem brains from both male and female, demented and non-demented patients (mean age ± SD 84.3±9.3 years). Using polyclonal antibodies to Aβ40 and 42, capillary and plaques positivity were assessed semiquantiatively on a four-point scale. Aβ42 deposits in capillaries correlated highly with both Aβ42 deposits in plaques and morphological AD criteria (CERAD, Braak stages, and NIA-Reagan-Institute criteria), while only a low correlation with CAA was observed. Aβ40 deposits in capillaries differed morphologically from Aβ42 ones: they were limited to capillary walls, were significantly less frequent in both capillaries and plaques compared to Aβ42 (P<0.01), and showed a low correlation with morphological AD criteria (P<0.05) and general CAA (P<0.01). By contrast, Aβ42 deposits were seen in the glia limitans rather than in capillary walls themselves, and showed high correlation with morphological AD criteria (P<0.01). These data indicate that CapCAA is characterized by Aβ42 deposits in pericapillary spaces or in the glia limitans. A low correlation between CAA and CapCAA, but high correlations between morphological AD criteria and CapCAA suggest different pathomechanisms for both types of CAA, and a close relation between CapCAA and AD pathology (both neuritic and plaque type). These data support the concept of a neuronal origin of Aβ via drainage from interstitial fluid from the central nervous system along basement membranes to capillaries.





Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer disease
- Aβ:
-
beta amyloid peptide
- Aβ 40/42 CapS:
-
score of deposits of Aβ 1–40/42 in capillaries
- Aβ 40/42 C:
-
number of Aβ 1–40/42 positive cortical vessels
- Aβ 40/42 PS:
-
score of deposits of Aβ 1–40/42 in plaques
- Aβ 40/42 TS:
-
total score of Aβ 1–40/42 deposits
- Aβ 40/42 Csev:
-
severity of Aβ 1–40/42 affection of cortical vessels
- Aβ 40/42 CS:
-
Aβ 1–40/42 cortical score
- Aβ 40/42 L:
-
percentage of Aβ 40/42 positive leptomeningeal vessels
- Aβ 40/42 Lsev:
-
severity of Aβ 40/42 affection of leptomeningeal vessels
- Aβ 40/42 LS:
-
Aβ 40/42 leptomeningeal score
- AβCTS:
-
Aβ cortical total score
- AβLTS:
-
Aβ leptomeningeal total score
- CAA:
-
cerebral amyloid angiopathy
- CAATS:
-
CAA total score
- CapCAA:
-
capillary CAA
- CERAD:
-
Consortium to Establish a Registry of Alzheimer’s Disease
- NFT:
-
neurofibrillary tangle
- NIA:
-
National Institute of Aging
- NIA-RI:
-
National Institute of Aging and Reagan Institute
- NP:
-
neuritic plaque
- SP:
-
senile plaque
- TS:
-
total score
References
Alonzo NC, Hyman BT, Rebeck GW, Greenberg SM (1998) Progression of cerebral amyloid angiopathy: accumulation of amyloid-beta40 in affected vessels. J Neuropathol Exp Neurol 57:353–359
Attems J, Jellinger KA (2004) Only cerebral capillary amyloid angiopathy correlates with Alzheimer pathology—a pilot study. Acta Neuropathol 107:83–90
Bergeron C, Ranalli PJ, Miceli PN (1987) Amyloid angiopathy in Alzheimer’s disease. Can J Neurol Sci 14:564–569
Braak H, Braak E (1991) Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol 82:239–259
Cadavid D, Mena H, Koeller K, Frommelt RA (2000) Cerebral beta amyloid angiopathy is a risk factor for cerebral ischemic infarction. A case control study in human brain biopsies. J Neuropathol Exp Neurol 59:768–773
Calhoun ME, Burgermeister P, Phinney AL, Stalder M, Tolnay M, Wiederhold KH, Abramowski D, Sturchler-Pierrat C, Sommer B, Staufenbiel M, Jucker M (1999) Neuronal overexpression of mutant amyloid precursor protein results in prominent deposition of cerebrovascular amyloid. Proc Natl Acad Sci USA 96:14088–14093
Castano EM, Prelli F, Soto C, Beavis R, Matsubara E, Shoji M, Frangione B (1996) The length of amyloid-beta in hereditary cerebral hemorrhage with amyloidosis, Dutch type. Implications for the role of amyloid-beta 1–42 in Alzheimer’s disease. J Biol Chem 271:32185–32191
Chalmers K, Wilcock GK, Love S (2003) APOE epsilon 4 influences the pathological phenotype of Alzheimer’s disease by favouring cerebrovascular over parenchymal accumulation of A beta protein. Neuropathol Appl Neurobiol 29:231–238
Dermaut B, Kumar-Singh S, De Jonghe C, Cruts M, Lofgren A, Lubke U, Cras P, Dom R, De Deyn PP, Martin JJ, Van Broeckhoven C (2001) Cerebral amyloid angiopathy is a pathogenic lesion in Alzheimer’s disease due to a novel presenilin 1 mutation. Brain 124:2383–2392
Duyckaerts C, Dickson DW (2003) Neuropathology of Alzheimer’s disease. In: Dickson DW (ed) Neurodegeneration. The molecular pathology of dementia and movement disorders. ISN Neuropath Press, Basel, pp 47–65
Ellis RJ, Olichney JM, Thal LJ, Mirra SS, Morris JC, Beekly D, Heyman A (1996) Cerebral amyloid angiopathy in the brains of patients with Alzheimer’s disease: the CERAD experience, Part XV. Neurology 46:1592–1596
Gravina SA, Ho L, Eckman CB, Long KE, Otvos L Jr, Younkin LH, Suzuki N, Younkin SG (1995) Amyloid beta protein (A beta) in Alzheimer’s disease brain. Biochemical and immunocytochemical analysis with antibodies specific for forms ending at A beta 40 or A beta 42(43). J Biol Chem 270:7013–7016
Hyman BT (1998) New neuropathological criteria for Alzheimer disease. Arch Neurol 55:1174–1176
Iwatsubo T, Odaka A, Suzuki N, Mizusawa H, Nukina N, Ihara Y (1994) Visualization of A beta 42(43) and A beta 40 in senile plaques with end-specific A beta monoclonals: evidence that an initially deposited species is A beta 42(43). Neuron 13:45–53
Iwatsubo T, Mann DM, Odaka A, Suzuki N, Ihara Y (1995) Amyloid beta protein (A beta) deposition: A beta 42(43) precedes A beta 40 in Down syndrome. Ann Neurol 37:294–299
Iwatsubo T, Saido TC, Mann DM, Lee VM, Trojanowski JQ (1996) Full-length amyloid-beta (1–42(43)) and amino-terminally modified and truncated amyloid-beta 42(43) deposit in diffuse plaques. Am J Pathol 149:1823–1830
Jellinger KA (2002) Alzheimer disease and cerebrovascular pathology: an update. J Neural Transm 109:813–836
Love S (2004) Contribution of cerebral amyloid angiopathy to Alzheimer’s disease. J Neurol Neurosurg Psychiatry 75:1–4
Mackic JB, Weiss MH, Miao W, Kirkman E, Ghiso J, Calero M, Bading J, Frangione B, Zlokovic BV (1998) Cerebrovascular accumulation and increased blood-brain barrier permeability to circulating Alzheimer’s amyloid beta peptide in aged squirrel monkey with cerebral amyloid angiopathy. J Neurochem 70:210–215
Mak K, Yang F, Vinters HV, Frautschy SA, Cole GM (1994) Polyclonals to beta-amyloid(1–42) identify most plaque and vascular deposits in Alzheimer cortex, but not striatum. Brain Res 667:138–142
Mann DM, Iwatsubo T (1996) Diffuse plaques in the cerebellum and corpus striatum in Down’s syndrome contain amyloid beta protein (A beta) only in the form of A beta 42(43). Neurodegeneration 5:115–120
Mann DM, Iwatsubo T, Ihara Y, Cairns NJ, Lantos PL, Bogdanovic N, Lannfelt L, Winblad B, Maat-Schieman ML, Rossor MN (1996) Predominant deposition of amyloid-beta 42(43) in plaques in cases of Alzheimer’s disease and hereditary cerebral hemorrhage associated with mutations in the amyloid precursor protein gene. Am J Pathol 148:1257–1266
Maruyama K, Ikeda S, Ishihara T, Allsop D, Yanagisawa N (1990) Immunohistochemical characterization of cerebrovascular amyloid in 46 autopsied cases using antibodies to beta protein and cystatin C. Stroke 21:397–403
Mirra SS, Heyman A, McKeel D, Sumi SM, Crain BJ, Brownlee LM, Vogel FS, Hughes JP, Belle G van, Berg L (1991) The Consortium to Establish a Registry for Alzheimer’s Disease (CERAD). Part II. Standardization of the neuropathologic assessment of Alzheimer’s disease. Neurology 41:479–486
Natte R, Boer WI de, Maat-Schieman ML, Baelde HJ, Vinters HV, Roos RA, Duinen SG van (1999) Amyloid beta precursor protein-mRNA is expressed throughout cerebral vessel walls. Brain Res 828:179–183
Oide T, Takahashi H, Yutani C, Ishihara T, Ikeda S (2003) Relationship between lobar intracerebral hemorrhage and leukoencephalopathy associated with cerebral amyloid angiopathy: clinicopathological study of 64 Japanese patients. Amyloid 10:136–143
Olichney JM, Hansen LA, Hofstetter CR, Grundman M, Katzman R, Thal LJ (1995) Cerebral infarction in Alzheimer’s disease is associated with severe amyloid angiopathy and hypertension. Arch Neurol 52:702–708
Olichney JM, Ellis RJ, Katzman R, Sabbagh MN, Hansen L (1997) Types of cerebrovascular lesions associated with severe cerebral amyloid angiopathy in Alzheimer’s disease. Ann N Y Acad Sci 826:493–497
Preston SD, Steart PV, Wilkinson A, Nicoll JA, Weller RO (2003) Capillary and arterial cerebral amyloid angiopathy in Alzheimer’s disease: defining the perivascular route for the elimination of amyloid beta from the human brain. Neuropathol Appl Neurobiol 29:106–117
Revesz T, Holton JL, Lashley T, Plant G, Rostagno A, Ghiso J, Frangione B (2002) Sporadic and familial cerebral amyloid angiopathies. Brain Pathol 12:343–357
Revesz T, Ghiso J, Lashley T, Plant G, Rostagno A, Frangione B, Holton JL (2003) Cerebral amyloid angiopathies: a pathologic, biochemical, and genetic view. J Neuropathol Exp Neurol 62:885–898
Revesz T, Ghiso J, Plant G, Holton JL, Frangione B (2003) Inherited amyloidosis and neurodegenerations in Familial British and Danish Dementia. In: Dickson DW (ed) Neurodegeneration. The molecular pathology of dementia and movement disorders. ISN Neuropath Press, Basel, pp 380–385
Roher AE, Kuo YM, Esh C, Knebel C, Weiss N, Kalback W, Luehrs DC, Childress JL, Beach TG, Weller RO, Kokjohn TA (2003) Cortical and leptomeningeal cerebrovascular amyloid and white matter pathology in Alzheimer’s disease. Mol Med 9:112–122
Saido TC, Iwatsubo T, Mann DM, Shimada H, Ihara Y, Kawashima S (1995) Dominant and differential deposition of distinct beta-amyloid peptide species, A beta N3(pE), in senile plaques. Neuron 14:457–466
Thal DR, Ghebremedhin E, Rub U, Yamaguchi H, Tredici KD, Braak H (2002) Two types of sporadic cerebral amyloid angiopathy. J Neuropathol Exp Neurol 61:282–293
Thal DR, Ghebremedhin E, Orantes M, Wiestler OD (2003) Vacular pathology in Alzheimer disease: correlation of cerebral amyloid angiopathy and arteriosclerosis/lipohyalinosis with cognitive decline. J Neuropathol Exp Neurol 62:1287–1301
Tian J, Shi J, Bailey K, Mann DM (2003) Negative association between amyloid plaques and cerebral amyloid angiopathy in Alzheimer’s disease. Neurosci Lett 352:137–140
Tian J, Shi J, Bailey K, Mann DM (2004) Relationships between arteriosclerosis, cerebral amyloid angiopathy and myelin loss from cerebral cortical white matter in Alzheimer’s disease. Neuropathol Appl Neurobiol 30:46–56
Vinters HV (1987) Cerebral amyloid angiopathy. A critical review. Stroke 18:311–324
Vinters HV, Secor DL, Pardridge WM, Gray F (1990) Immunohistochemical study of cerebral amyloid angiopathy. III. Widespread Alzheimer A4 peptide in cerebral microvessel walls colocalizes with gamma trace in patients with leukoencephalopathy. Ann Neurol 28:34–42
Vonsattel JP, Myers RH, Hedley-Whyte ET, Ropper AH, Bird ED, Richardson EP Jr (1991) Cerebral amyloid angiopathy without and with cerebral hemorrhages: a comparative histological study. Ann Neurol 30:637–649
Weller RO, Nicoll JA (2003) Cerebral amyloid angiopathy: pathogenesis and effects on the ageing and Alzheimer brain. Neurol Res 25:611–616
Weller RO, Massey A, Newman TA, Hutchings M, Kuo YM, Roher AE (1998) Cerebral amyloid angiopathy: amyloid beta accumulates in putative interstitial fluid drainage pathways in Alzheimer’s disease. Am J Pathol 153:725–733
Weller RO, Massey A, Kuo YM, Roher AE (2000) Cerebral amyloid angiopathy: accumulation of A beta in interstitial fluid drainage pathways in Alzheimer’s disease. Ann N Y Acad Sci 903:110–117
Xu D, Yang C, Wang L (2003) Cerebral amyloid angiopathy in aged Chinese: a clinico-neuropathological study. Acta Neuropathol 106:89–91
Yamada M, Tsukagoshi H, Otomo E, Hayakawa M (1987) Cerebral amyloid angiopathy in the aged. J Neurol 234:371–376
Zekry D, Duyckaerts C, Belmin J, Geoffre C, Moulias R, Hauw JJ (2003) Cerebral amyloid angiopathy in the elderly: vessel walls changes and relationship with dementia. Acta Neuropathol 106:367–373
Zlokovic BV, Ghiso J, Mackic JB, McComb JG, Weiss MH, Frangione B (1993) Blood-brain barrier transport of circulating Alzheimer’s amyloid beta. Biochem Biophys Res Commun 197:1034–1040
Acknowledgements
The authors thank Mrs. Veronika Rappelsberger and Mrs. Barbara Weidinger for excellent laboratory work, Erich Mitter-Ferstl, PhD, for secretarial and computer work, and Prof. Roy Weller, Southampton, for critical remarks. Part of the study was supported by the Society for the Support of Research in the Field of Experimental Neurology and by Consultatio GmbH, both Vienna, Austria.
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article can be found at http://dx.doi.org/10.1007/s00401-004-0866-7
Rights and permissions
About this article
Cite this article
Attems, J., Lintner, F. & Jellinger, K.A. Amyloid β peptide 1–42 highly correlates with capillary cerebral amyloid angiopathy and Alzheimer disease pathology. Acta Neuropathol 107, 283–291 (2004). https://doi.org/10.1007/s00401-004-0822-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-004-0822-6